
↑食藥署於4月17日舉辦「藥品守護天使-藥品AI新世代 就是藥Good」記者會。

↑食藥署「西藥、醫療器材、化粧品許可證查詢系統」。
【本刊訊】
隨著民眾用藥意識高漲,食品藥物管理署於4月17日舉辦「藥品守護天使-藥品AI新世代 就是藥Good」記者會,說明藥品已進入AI新世代,民眾可透過官方查詢系統取得藥品所有的資訊。
藥品的製造生產過程食藥署如何進行把關?民眾該如何確定取得的藥品是通過食藥署核准的?食藥署藥品組科長林意筑表示,藥品進入AI新世代,即指用藥可近性(Accessibility)與智慧科技(Intelligence)的結合。食藥署從藥品非臨床試驗、臨床試驗、上市審查、生產製造、運銷至上市後監控,全面強化智慧科技之運用,以提升藥品可近性。
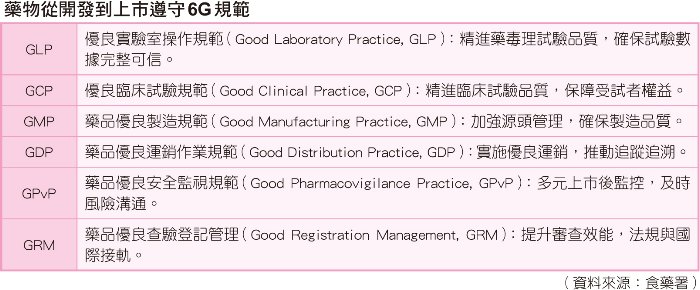
藥品從開發到上市,大約要十年的時間,開發者、研發者都要遵守6G規範,包括優良實驗室操作規範(Good Laboratory Practice, GLP)、優良臨床試驗規範(Good Clinical Practice, GCP)、藥品優良製造規範(Good Manufacturing Practice, GMP)、藥品優良運銷作業規範(Good Distribution Practice, GDP)、藥品優良安全監視規範 (Good Pharmacovigilance Practice, GPvP)、藥品優良查驗登記管理(Good Registration Management, GRM)等,持續確保藥品品質、安全及療效,以守護全民用藥安全。
食藥署表示,通過食藥署核准的藥品,會有許可證字號,藥師可教導民眾在藥品的外盒包裝、標籤及說明書(仿單)都可以看到許可證字號,民眾可於食藥署「西藥、醫療器材、化粧品許可證查詢系統」(http://www.fda.gov.tw/mlms/H0001.aspx)輸入藥品「許可證字號」或中、英文「品名」,即可查詢藥品、仿單、藥盒外觀、成分等藥品資訊,另外也可查詢藥品之限制項目,包括管制進口、新藥監視、限期註銷、限醫院使用、監視中新藥、原料未取得PIC/S GMP核准函不得製造等用藥資訊,對自我用藥品項有更深的概念。
回首頁 |